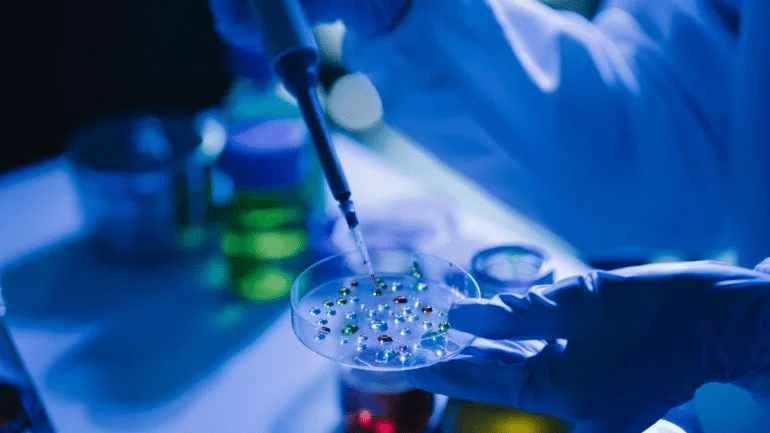
We Provide Specialized Pharmaceutical Services
Empowering pharmaceutical companies across the globe, we deliver specialized consulting, research, and analytical services. We ensure the highest standards of quality, testing, and regulatory compliance, driving innovation to meet the demands of a dynamic industry.
R&D Services
Our R&D lab drives innovation through formulation and method development that meets strict standards.
Regulatory Support
Expert pharmaceutical consulting to ensure quality and regulatory compliance.
About us


290
Analytical Tests Performed
65
R&D Projects Completed
35
Client Excellence
22
Global Supplier Partnerships
We are trusted by World's leading companies
At our core is a distinguished and multidisciplinary team of professionals, bringing together PhD-level scientists, seasoned regulatory specialists, and veteran industry consultants. This collective expertise is wholly dedicated to delivering exceptional service and in-depth scientific support, specifically tailored to the rigorous demands of the pharmaceutical industry. We understand that success in this field is non-negotiable, which is why we operate within a culture of uncompromising quality.
Our commitment to compliance is absolute. We ensure strict adherence to global regulatory standards, such as GMP and GLP, in every facet of our consulting, research, and analytical services. From initial study design to final report generation, our processes are meticulously documented and managed through a robust quality system, guaranteeing that the data we deliver is not only accurate but also fully defensible.
We work with a wide range of clients across the pharmaceutical and biotechnology sectors, including research organizations, clinical facilities, and pharmaceutical manufacturers. Our partners trust us for high-quality consulting, analytical testing, and R&D services tailored to their regulatory and scientific needs.
We offer a full range of services including consulting, laboratory research and development, and analytical testing. Our capabilities cover QA/QC testing, stability studies, regulatory compliance support, and scientific insight tailored to your product development goals.
Quality, safety, and scientific integrity are embedded into every step of our process. We develop and validate each service to meet rigorous regulatory requirements, ensuring our clients’ products meet international standards and market expectations.
We believe in building long-term partnerships rooted in trust, transparency, and shared goals. Our client-focused approach means we adapt our solutions to your needs and support you at every stage from early R&D through regulatory approval and beyond.
Different types of high-quality
services department
Pharmaceutical Project Support
We support pharmaceutical projects with API documentation and technical insight to ensure quality, compliance, and successful formulation.
Research & Development
We offer innovative R&D services in formulation, process optimization, and tech transfer to support safe, effective pharmaceutical products.
Customized Laboratory Services
We provide tailored lab services from contract research to pilot-scale support designed to deliver insights and accelerate development.
Regulatory compliance
Regulatory compliance support tailored to the pharmaceutical industry ensuring all products meet Canadian standards through expert guidance, documentation, and audit readiness.
wide range of brand-name
Includes medications that have been developed, tested, and approved by reputable companies, providing safe and effective treatment options for a variety of medical conditions.
medical devices
Devices and equipment used for diagnosing, treating, or preventing diseases and medical conditions.
LET'S TALK ...
Our excellent team is ready to help you achieve your business objectives.
We are working with
World's leading companies





Latest news & Blogs